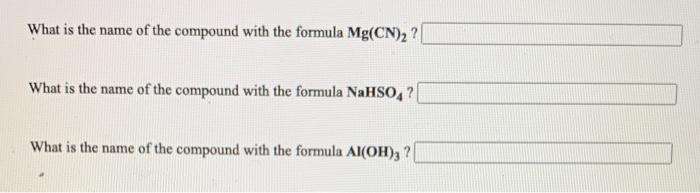
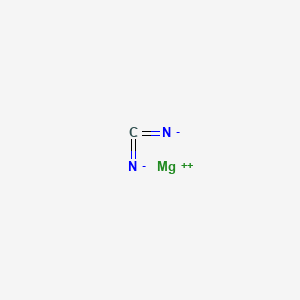
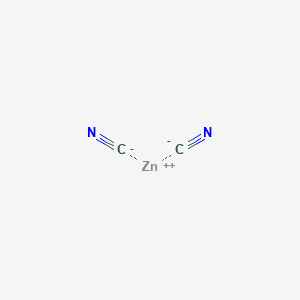
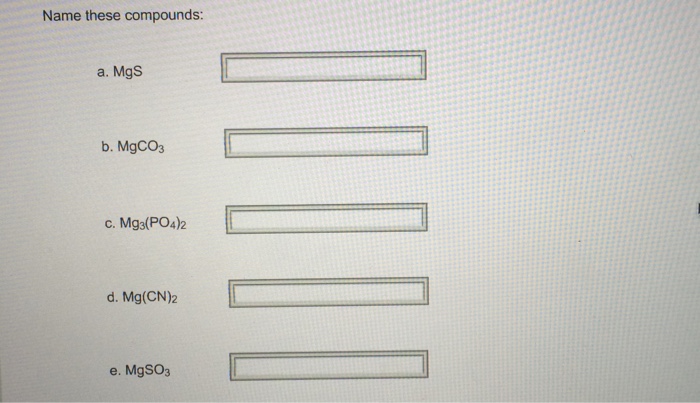
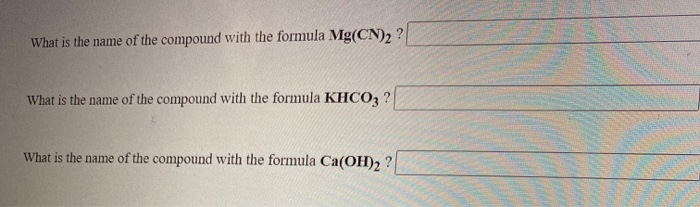
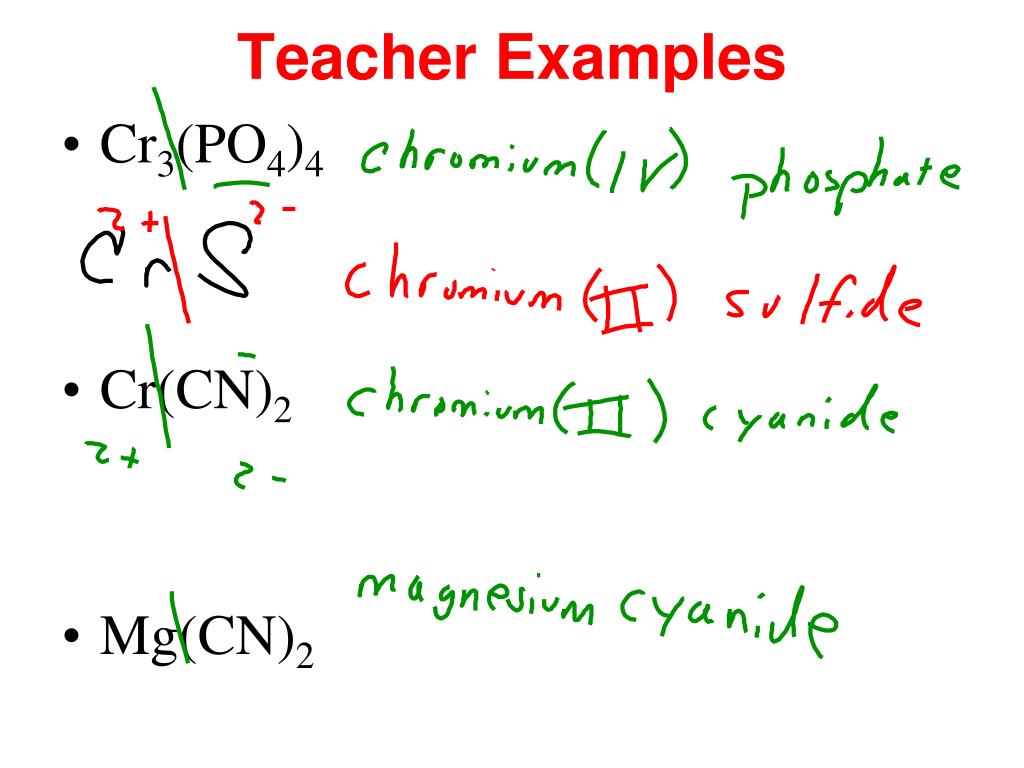
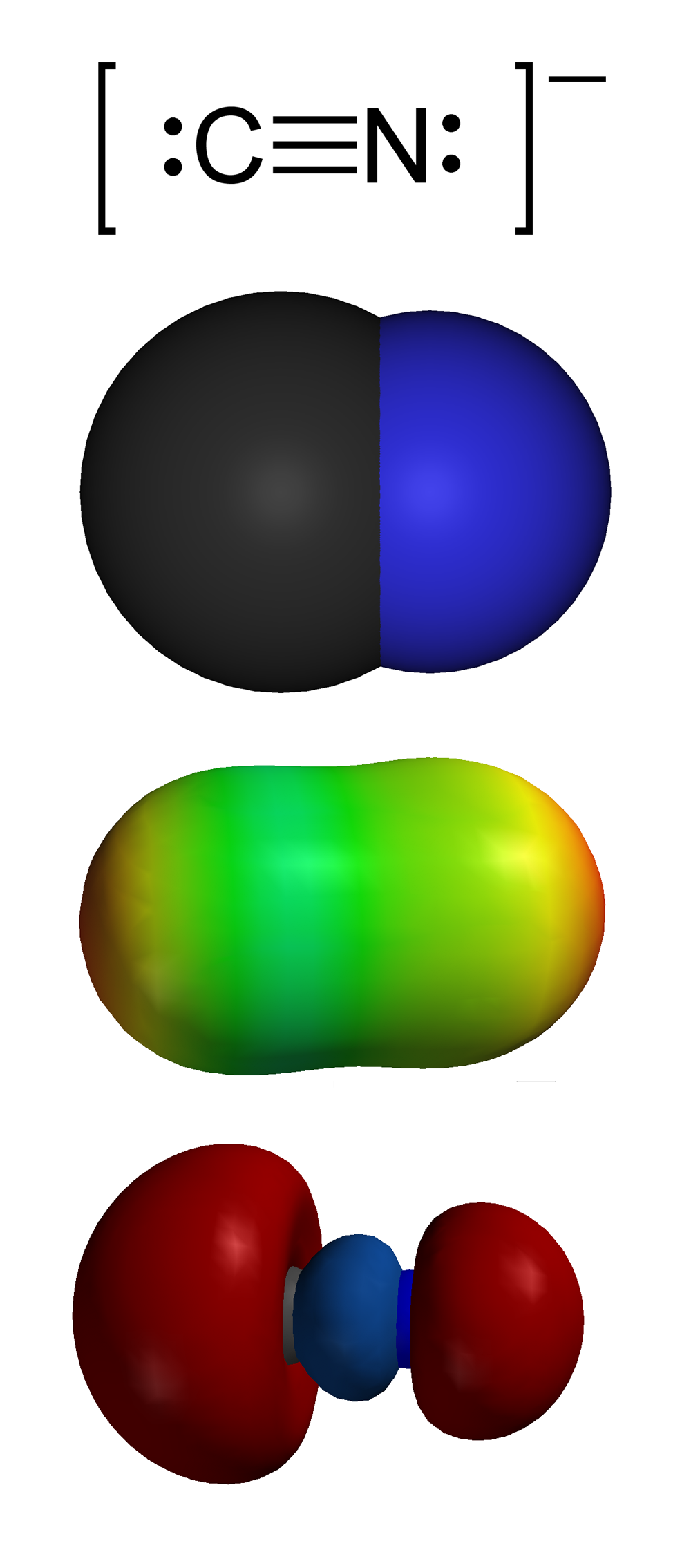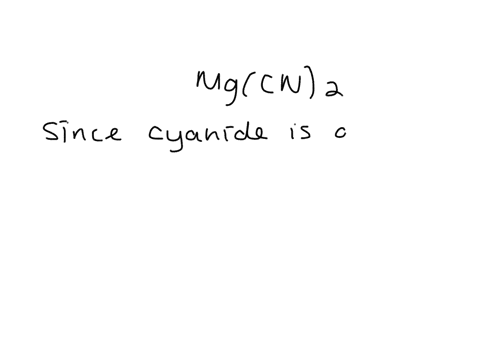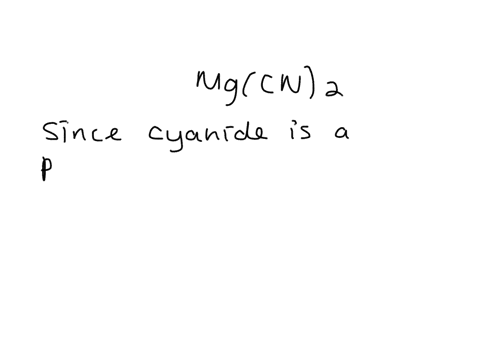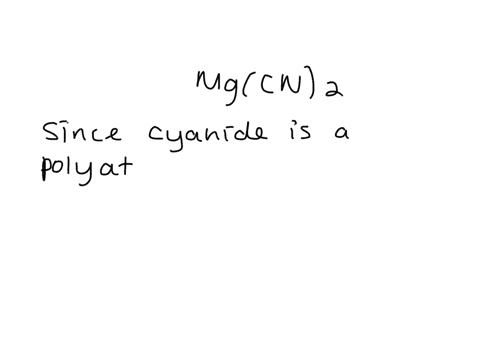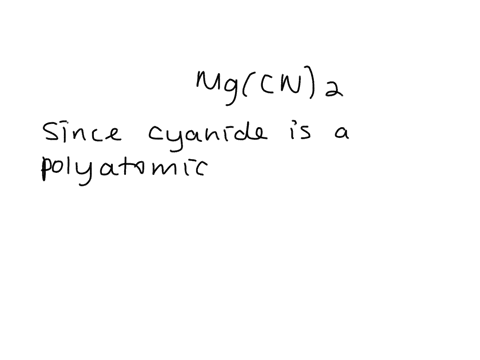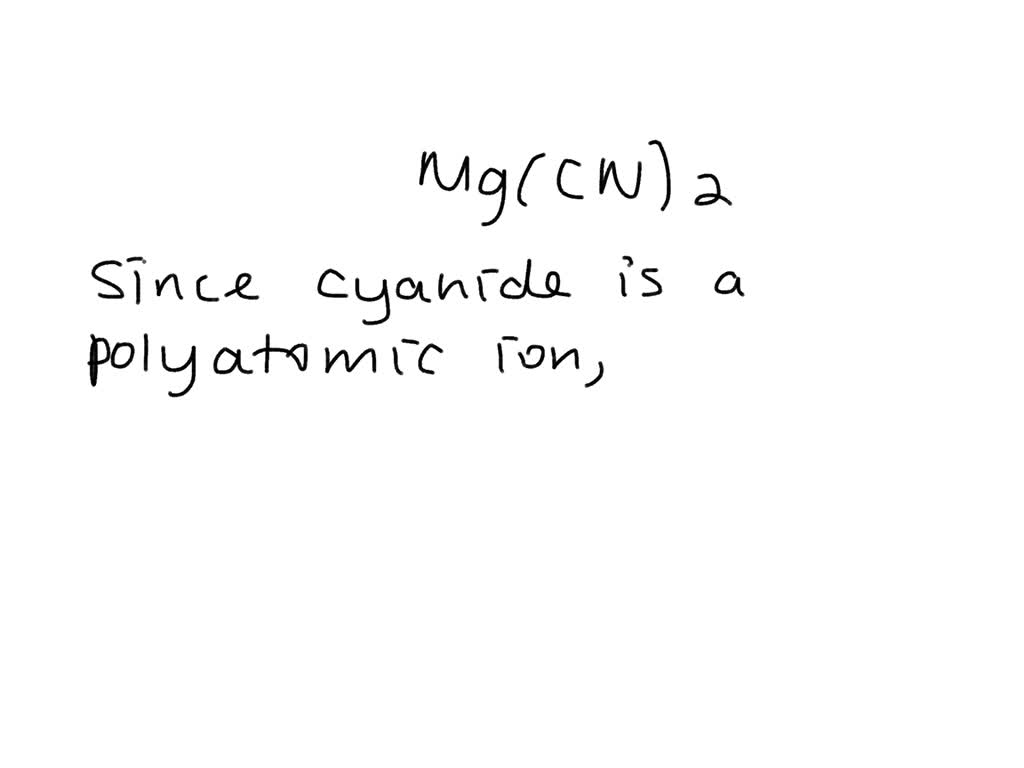SOLVED: Give the name of the polyatomic ions found in Mg(CN)2. A. carbon nitride B. dicyanide C. dicarbon dinitride D. cyanide

SOLVED: Give the name of the polyatomic ions found in Mg(CN)2. A. carbon nitride B. dicyanide C. dicarbon dinitride D. cyanide

SOLVED: 24. Assuming reactions between the following pairs of elements, which pair is most likely to form a covalent compound? A) lithium and iodine B) sodium and oxygen C) calcium and chlorine