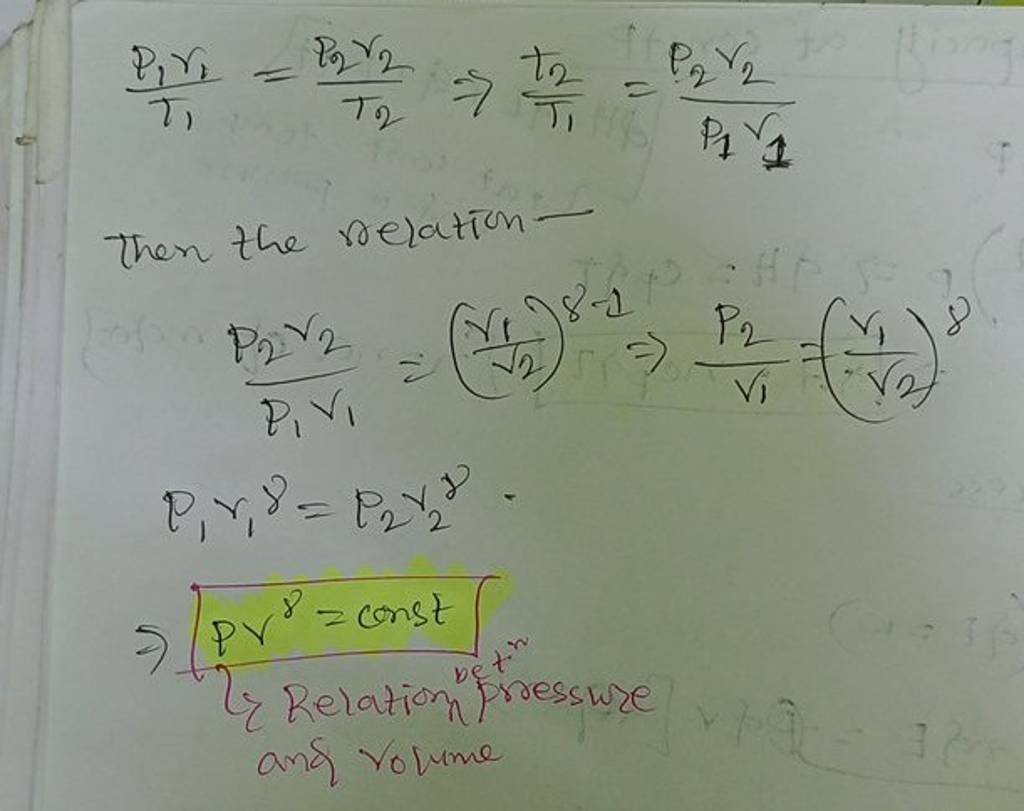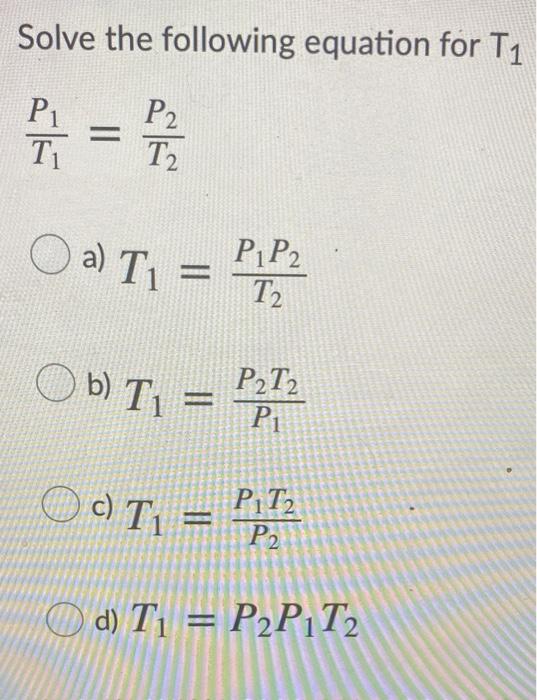Two thermally insulated vessels 1 and 2 are filled with air temperatures (T1, T2), volume (V1, V2) and pressure (P1, P2) respectively. If the valve joining two vessels is opened, the temperature
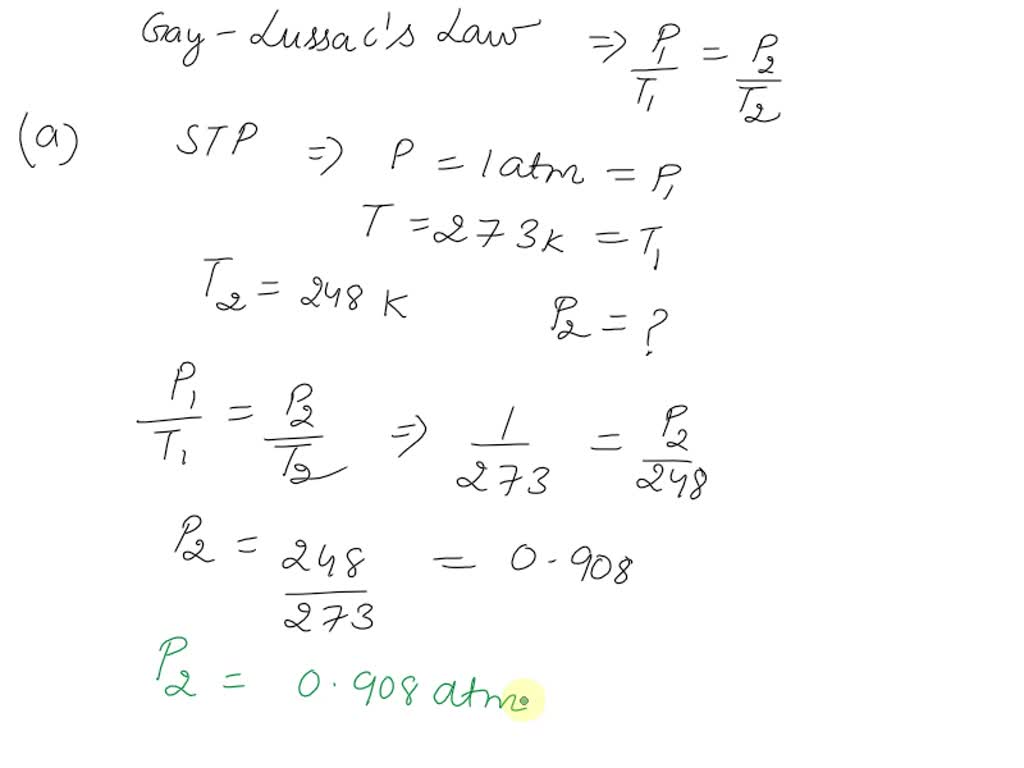
SOLVED: Gay-Lussac's Law: P1T2 = P2T1 or P1/T1 = P2/T2 T must be in Kelvin (K = 273 + C) changed to 127°C while the volume remains constant. 2. A gas initially

Changes in average P2/P1, average t2/t1 and average V/P1 for different... | Download Scientific Diagram

C2,C3,K1,K3,L1,L2,N1,N2,R1,R2,M1,M2,P1,P2,T, Türü Yetki Belgesi Sahibi Firmalara Önemli Duyuru! - MUĞLA TİCARET VE SANAYİ ODASI | MUTSO
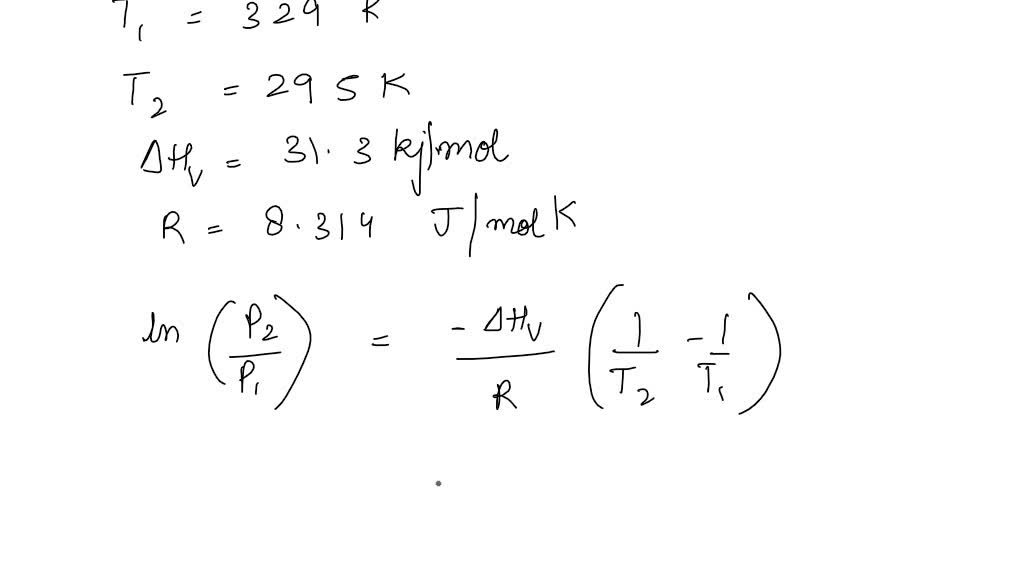
SOLVED: Determine the vapor pressure of your unknown liquid at 295 K using the Clausius-Clapeyron equation. Show all of your work with units. Next to each appropriate value used, put P1, P2,





![ANSWERED] One mole of a real gas is subjected to heating at constant - Kunduz ANSWERED] One mole of a real gas is subjected to heating at constant - Kunduz](https://media.kunduz.com/media/sug-question-candidate/20210603014324956133-3161513.jpg)